How can we utilise PRF in dentistry?
PRF has a wide variety of use in dental surgery, dental extraction, dental implantology, periodontal regeneration, bone grafting.
PRF is an autologous fibrin-based living biomaterial, derived from human blood. PRF accelerates soft tissue healing and bone regeneration which are the fundamentals of successful and long lasting dental implant treatment.
The purpose of PRF treatment is to extract from a patient’s blood sample the key elements and to prepare it in a clinically usable form such as a membrane or plug (A-PRF, L-PRF) or injectable liquid (i-PRF).
PLATELETS, LEUKOCYTES and STEM CELLS are the key active elements trapped in the fibrin matrix which will be connected with local tissues (bone and soft tissue) to accelerate new blood vessel formation and enhance its healing and regeneration potential.
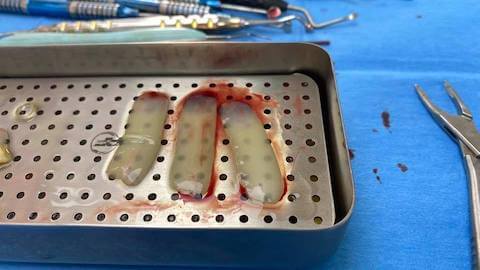
Physical application of PRF

PRF (A-PRF or L-PRF) can either be used as
- a clot
- membrane
- injectable liquid (i-PRF)
- plug, or
- cut up in fragments.
Use of PRF in stand-alone, additive, or in combination therapies
Stand-alone therapies
Typical stand-alone therapies include using the fibrin plug or membrane as a filler material in extraction sockets to prevent complications and to enhance socket healing (Choukroun, et al., 2006).
PRF can also be used as a protective barrier membrane to seal off and promote healing of oroantral communications following extractions; to close a palatal connective tissue harvesting site; or as sole grafting material in sinus floor elevations.

Additive therapies
PRF (membrane or liquid) can be added or mixed to bone substitutes such as xenograft or biphasic calcium phosphate (BCP) to enhance the formation of new bone.

Combination therapies
The membrane is often used in combination with other biomaterials in bone augmentation and grafting sites as a graft material or barrier membrane (Hamzacebi, et al., 2015).
The purpose of PRF is to activate and facilitate the healing and regenerative capacity of the host tissue, by providing a strong fibrin scaffold, major growth factors, and allowing space for tissue regeneration.
Using PRF as a protective barrier on bone graft sites helps avoid perforations of the weakened gingival tissues and prevent associated contamination of the bone graft below.


PRF course with the inventor Dr. Joseph Choukroun
25-26th March, 2022 London
- Dr. Choukroun’s upcoming Platelet Rich Fibrin -PRF- course is an enlightenment on the biological and mechanical conditions for long term stability and success of bony and soft tissue management.
- Learn about PRF use in dental implantology, oral surgery, bone graft. Prepare PRF products on the hands-on session, practice phlebotomy and start utilising PRF from the next day in your dental practice.

The use of Choukroun’s PRF treatment enables local delivery of a fibrin matrix, cells, growth factors, and proteins that provide unique biological properties and cues for promoting new blood vessel formation, and accelerating wound healing and tissue regeneration, while at the same time reducing adverse events. The benefits of PRF treatment in wound and bone healing, its antibacterial and antihemorrhagic effects, the low risks with its use, and the availability of easy and low-cost preparation methods should encourage more clinicians to adopt this technology in their practices for the benefit of their patients
If you liked this article check out our social media pages for more content.
References:
Drs. Johan Hartshorne and Howard Gluckman: A comprehensive clinical review of platelet-rich fibrin (PRF) and its role in promoting tissue healing and regeneration: part 2
Choukroun J, Diss A, Simonpieri A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part V: histologic evaluations of PRF effects on bone allograft maturation in sinus lift. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 101:299-303.
Choukroun J, Diss A, Simonpieri A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e56-e60


